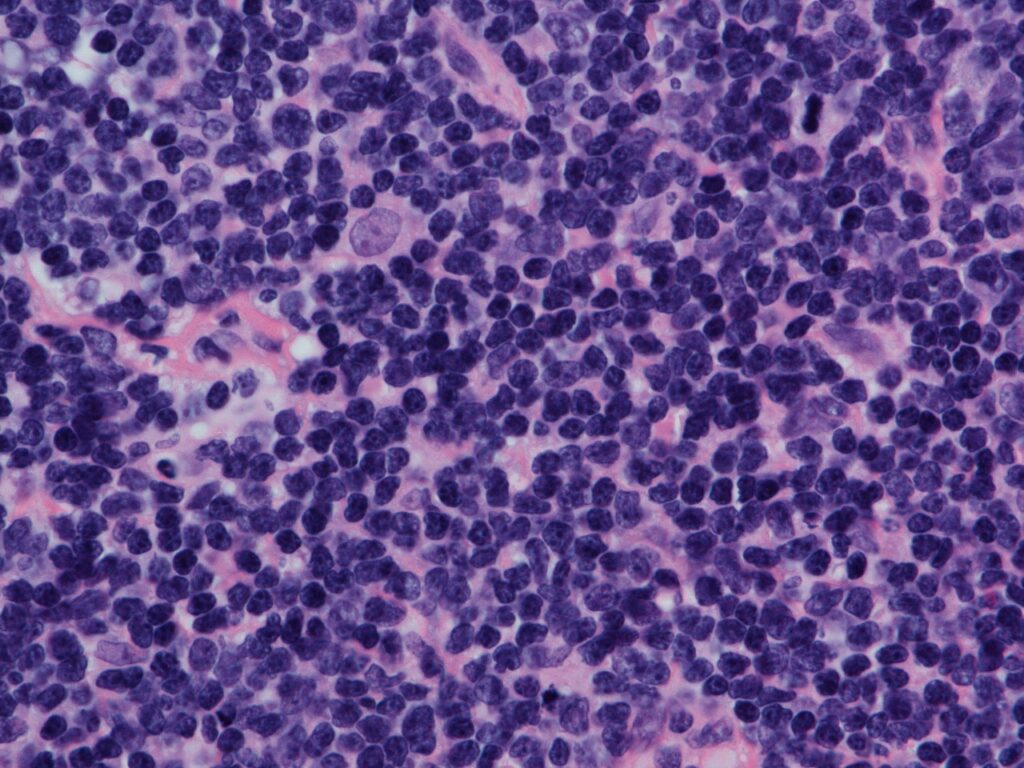
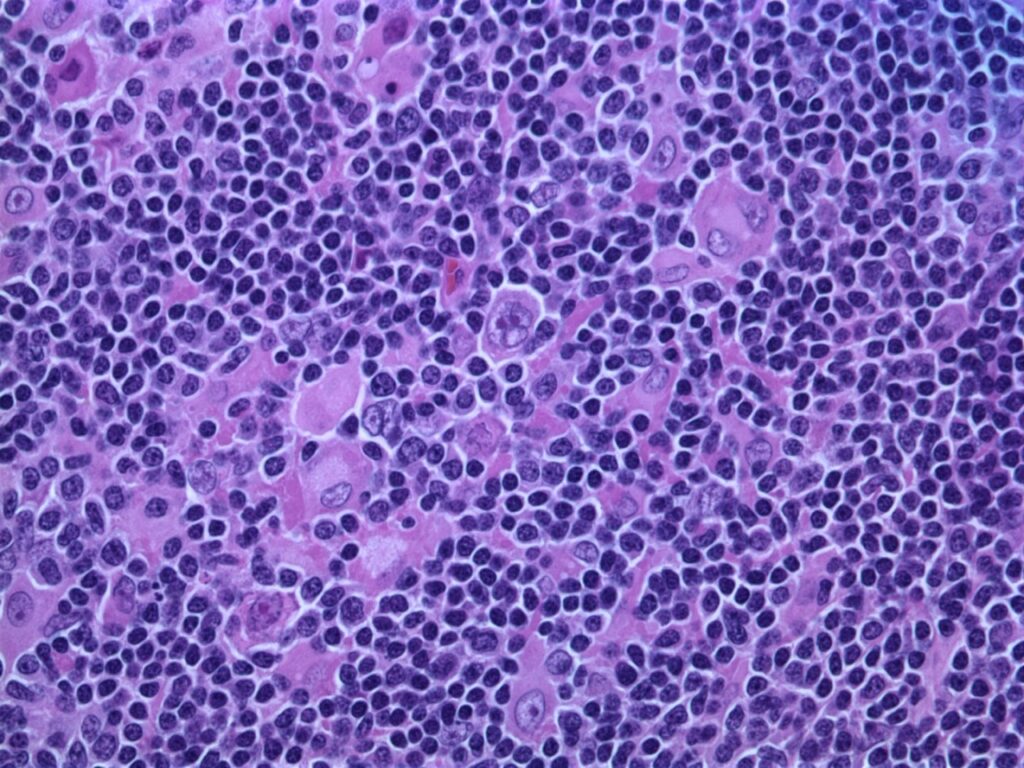
Janssen submits application to EMA for mantle cell lymphoma therapy approval
The Janssen Pharmaceutical Companies of Johnson & Johnson has submitted a Type II variation application seeking approval for the new indication of Imbruvica (ibrutinib) plus bendamustine and rituximab (BR) combination from the European Medicines Agency (EMA).
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.