Sandoz seeks EMA approval for EirGenix’s trastuzumab biosimilar
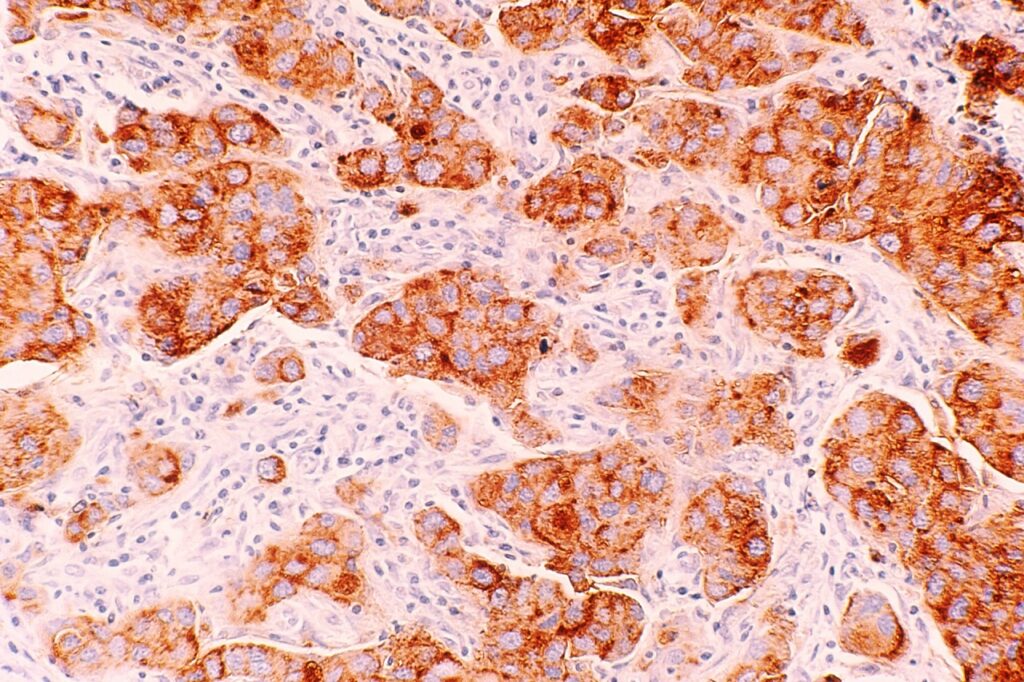
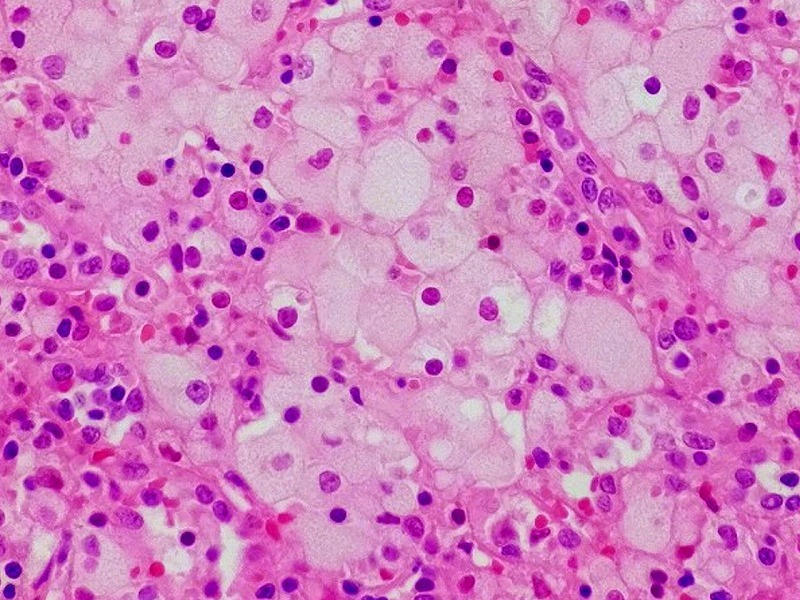
Novartis’ subsidiary Sandoz has submitted a marketing authorisation application to the European Medicines Agency (EMA) for the EirGenix-developed proposed trastuzumab biosimilar drug (150 mg).
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.