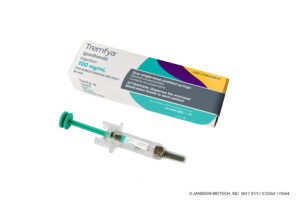
Janssen’s Tremfya meets primary endpoint in phase 3 psoriasis trial
The phase 3 trial, called ECLIPSE, enrolled 1048 patients to evaluate the efficacy and safety of Tremfya in comparison to Cosentyx. While Tremfya is an interleukin (IL)-23-targeted biologic
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.