The US Food and Drug Administration (FDA) has accepted Amneal Pharmaceuticals’ new drug application (NDA) for IPX203 to treat Parkinson’s disease (PD).
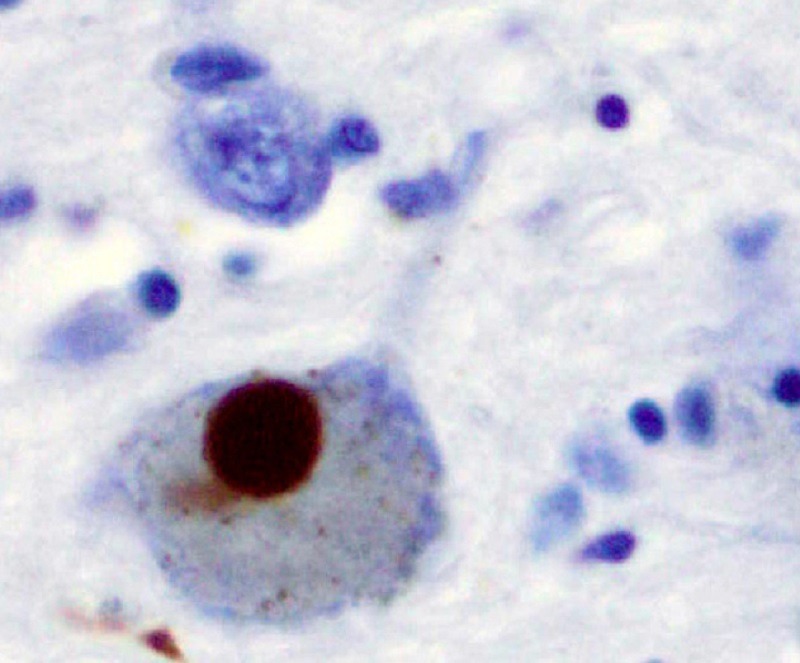
A Lewy body (stained brown) in a brain cell of the substantia nigra in Parkinson's disease. Credit: Marvin 101/ commons.wikimedia.org.
Subscribe to our email newsletter
The new, oral formulation of CD/LD extended-release capsules, IPX203 features immediate-release granules and extended-release beads.
Findings from the pivotal Phase III RISE-PD clinical trial showed that the extended-release formulation of IPX203 has provided significantly more “Good On” time, and less “Off” time, compared with immediate-release CD/LD.
The double-dummy, randomised, active-controlled, double-blind, multicentre, parallel-group RISE-PD trial assessed the safety and efficacy of IPX203 CD/LD extended-release capsules compared with immediate-release CD/LD to treat PD patients who have motor fluctuations.
The trial included 506 PD patients and was conducted at 105 clinical sites in the US, European countries, including Italy, Czechia, France, Spain, Germany, Poland, and the UK.
Amneal Specialty chief commercial officer Gustavo Pesquin said: “The FDA filing acceptance of IPX203 marks another important milestone for Amneal as we strive to improve the lives and care of people living with Parkinson’s disease.
“We look forward to engaging in conversations with the FDA as we advance the application.
“We believe the data in our RISE-PD study supports the important benefit IPX203 can offer to this community by providing longer duration of symptom control with the benefit of fewer doses.”
The company expects IPX203 to provide a longer duration of symptom control for PD patients with less frequent dosing compared with IR CD/LD treatment.
The regulatory body has allotted a Prescription Drug User Fee Act (PDUFA) action date of 30 June 2023 to complete the NDA evaluation for IPX203.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.