Forge Biologics has received priority medicines (PRIME) designation from the European Medicines Agency (EMA) for its drug candidate FBX-101 to treat patients with Krabbe disease.
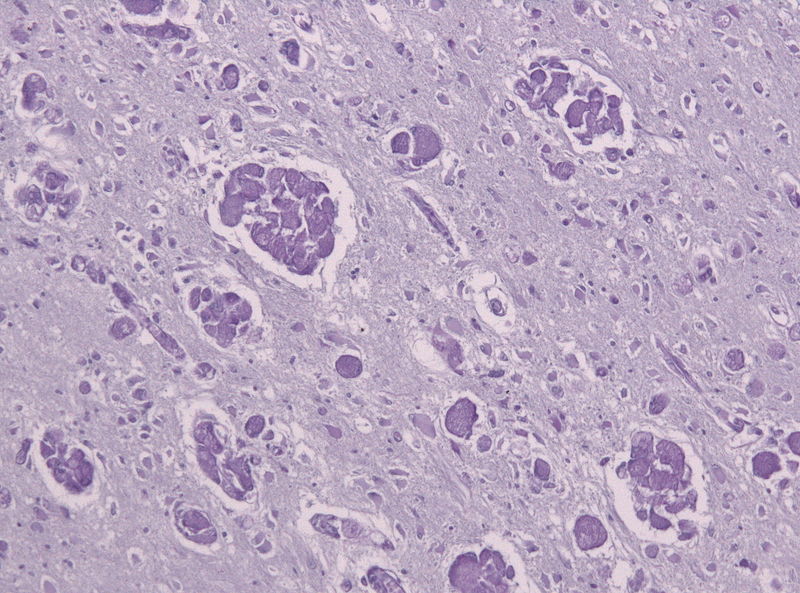
The RESKUE trial has been designed to study the safety and efficacy of FBX-101 in Infantile Krabbe disease patients. Credit: Jensflorian / commons.wikimedia.org.
Subscribe to our email newsletter
The adeno-associated viral serotype rh10 (AAVrh10) gene therapy FBX-101 is delivered intravenously after hematopoetic stem cell transplant (HSCT) infusion.
The designation for this lead adeno-associated virus (AAV) drug candidate has been granted after review of the safety and efficacy results obtained from the ongoing Phase I/II RESKUE trial.
RESKUE is a nonblinded, non-randomised dose escalation study of intravenous AAVrh10 and has been designed to study the safety and efficacy of FBX-101 in Infantile Krabbe disease patients.
The trial results demonstrated that systemically delivered FBX-101 administered after HSCT is safe and well-tolerated in these patients.
In the low dose group, patients demonstrated restoration of GALC enzyme activity, reduced psychosine, and reported encouraging normal myelination signs of brain white matter.
Motor development also improved in these patients compared to untreated Krabbe patients or patients treated with HSCT alone.
Forge Biologics Regulatory Affairs and Quality senior vice-president Christopher Shilling said: “We are grateful to the EMA for recognising FBX-101 as a potentially transformative medicine for patients living with this life-threatening and devastating disease.
“Through the enhanced interactions with the EMA granted by the PRIME designation, we will advance and expedite the development of FBX-101 as the leading worldwide gene therapy for patients with Krabbe disease.”
In animal models, FBX-101 demonstrated to functionally correct the central and peripheral neuropathy associated with Krabbe.
Treatment with the drug also improved gross motor outcomes, and prolonged lifespan in these models.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.