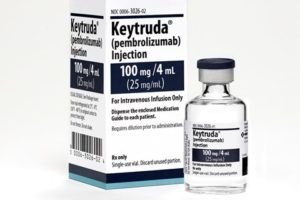
EC approves two new regimens of Merck’s KEYTRUDA for metastatic or unresectable recurrent HNSCC
This approval is based on findings from the pivotal Phase 3 KEYNOTE-048 trial, in which KEYTRUDA, compared with standard treatment (cetuximab with carboplatin or cisplatin plus 5-FU), demonstrated
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.