GSK gets Chinese approval for shingles vaccine Shingrix
Shingrix is a non-live and recombinant subunit adjuvanted vaccine, which will be administered intramuscularly in two doses. Shingles is caused by the reactivation of the varicella zoster virus
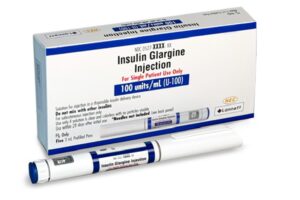
Sunshine Lake Pharma, Lannett and its wholly owned subsidiary Lanexa Biologics have received approval from the US Food and Drug Administration (FDA) for Langlara (insulin glargine-aldy), an interchangeable biosimilar of Lantus (insulin glargine).
This revolutionary platform empowers scientists in the pharmaceutical industry to generate novel drug-like compounds by combining powerful methods for molecule generation, ADMET prediction, and high-throughput ligand-protein interaction prediction.