Dr. Reddy’s Laboratories announces acquisition of ANDA portfolio
The portfolio includes more than 30 generic injectable products. These products will require to be technology transferred and could be launched within the next one to two years.
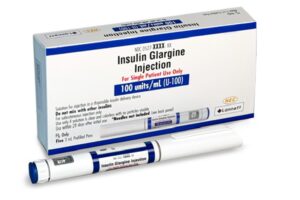
Sunshine Lake Pharma, Lannett and its wholly owned subsidiary Lanexa Biologics have received approval from the US Food and Drug Administration (FDA) for Langlara (insulin glargine-aldy), an interchangeable biosimilar of Lantus (insulin glargine).
The intended clinical trial will be a proof-of-concept study combining Novo Nordisk’s semaglutide (GLP-1 analogue) and Gilead’s cilofexor (FXR agonist) and firsocostat (ACC inhibitor) for the treatment of
The Phase 1/2 dose-rising trial will evaluate the safety and preliminary efficacy of NanoDoce for patients with high-risk non-muscle invasive bladder cancer (NMIBC) and muscle invasive bladder cancer