Arch Oncology raises $50m Series B financing
The Company plans to use the proceeds from this financing to advance its anti-CD47 antibody AO-176’s ongoing Phase 1 clinical trial in select solid tumors, as well as
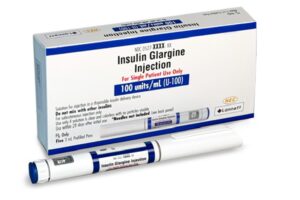
Sunshine Lake Pharma, Lannett and its wholly owned subsidiary Lanexa Biologics have received approval from the US Food and Drug Administration (FDA) for Langlara (insulin glargine-aldy), an interchangeable biosimilar of Lantus (insulin glargine).
“Today’s submission brings us one step closer to our goal of improving treatment outcomes for people newly diagnosed with multiple myeloma,” said José Antonio Burón Vidal, VP Medical