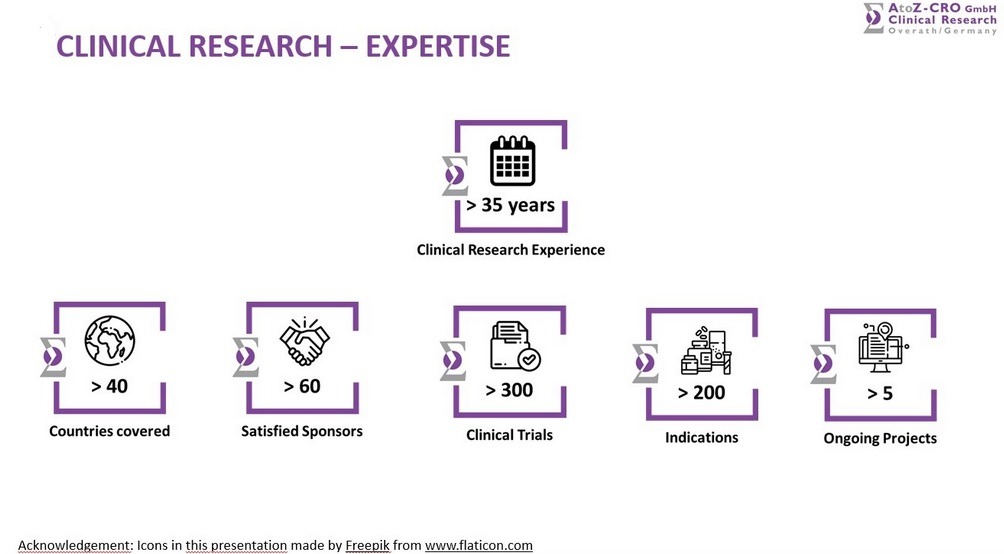
From its head office in Overath, Germany, AtoZ-CRO manages and coordinates clinical studies of phase I to IV and post-marketing studies for national and international clients. The company has successfully closed rescue studies and feasibility studies as well.
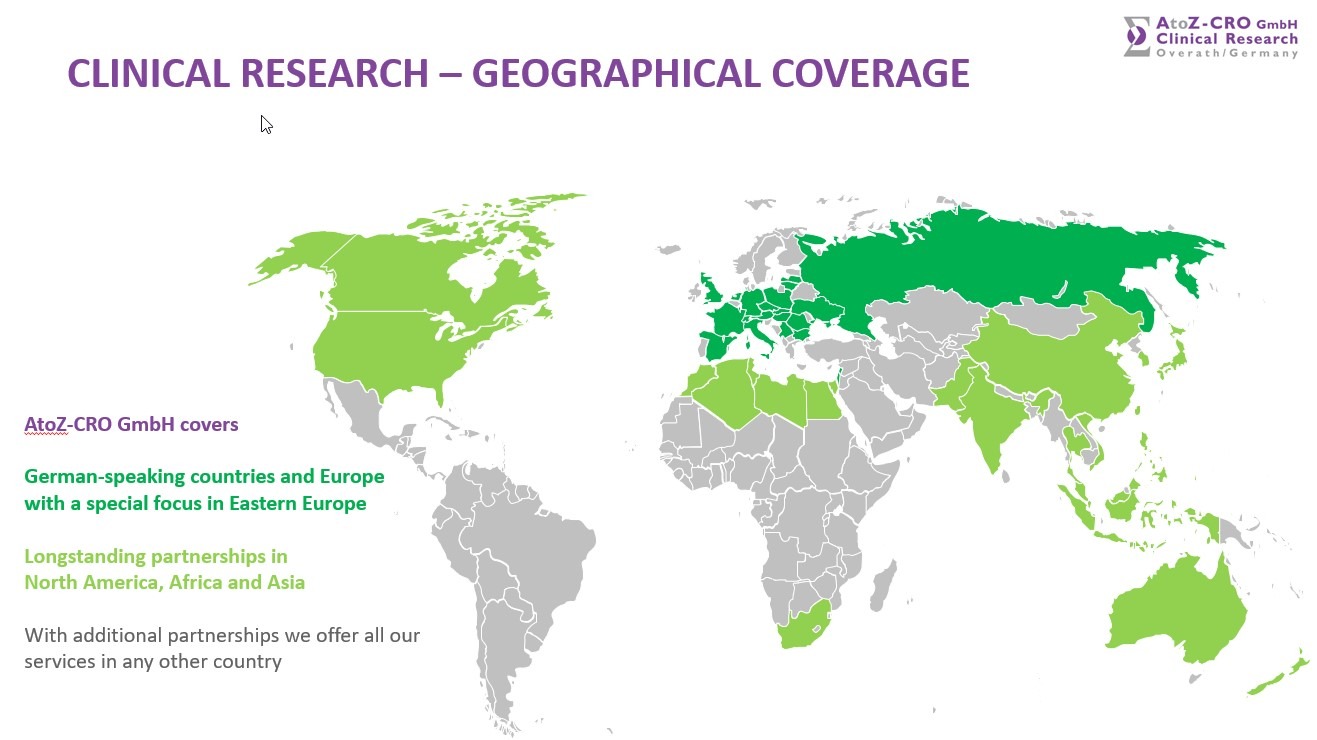
AtoZ-CRO GmbH covers German-speaking countries and Europe with a special focus in Eastern Europe. We have longstanding partnerships in North America, Asia and Africa, with additional partnerships we offer all our services in any other country.
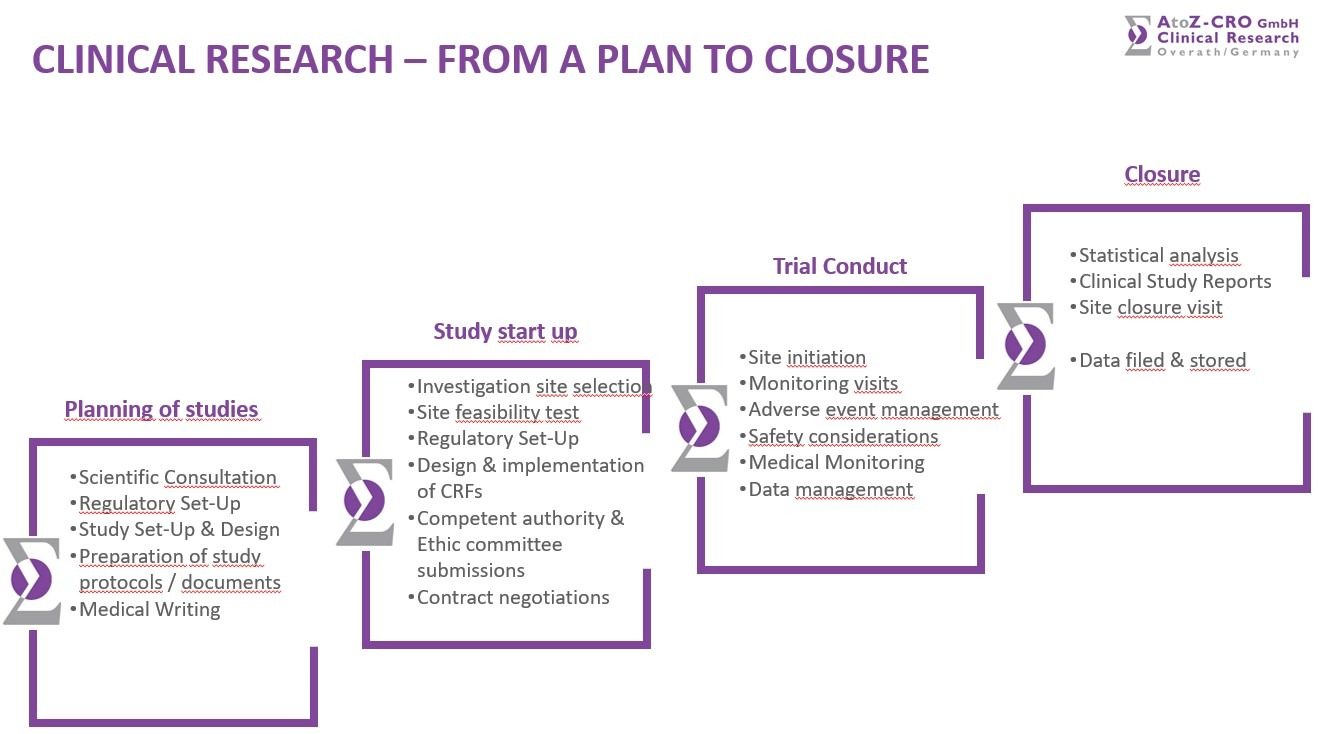
The company forms a team of highly experienced professionals and medical specialists ensuring high professional standards in clinical study management. Its services comprise the planning of clinical studies, preparation of study protocols, design and implementation of (e)CRFs and Regulatory Affairs issues.
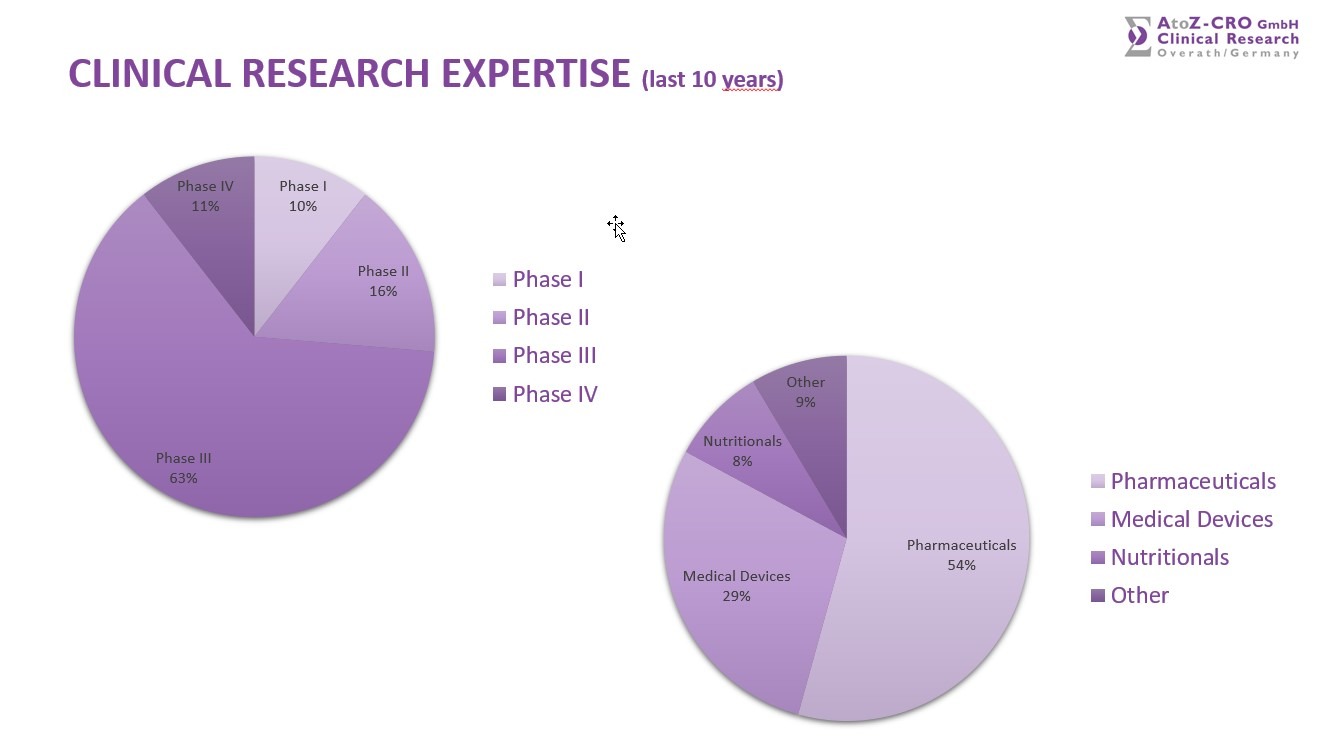
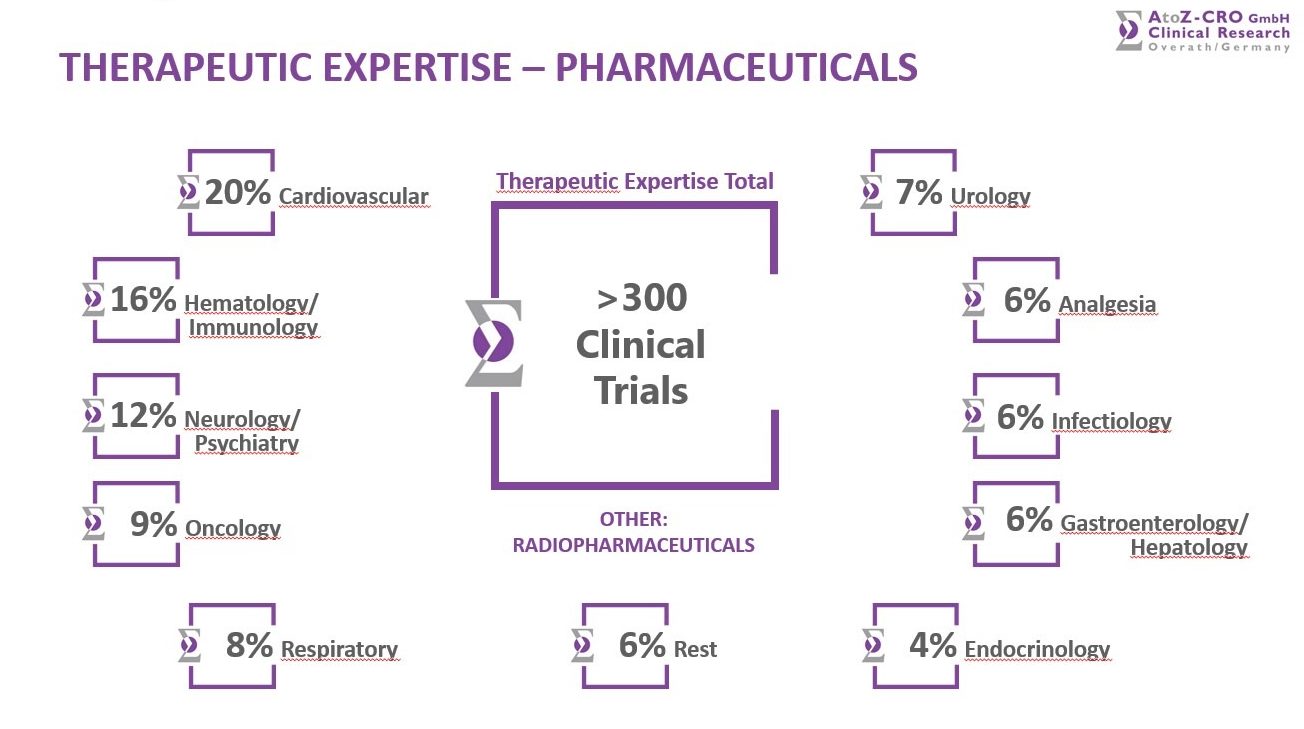
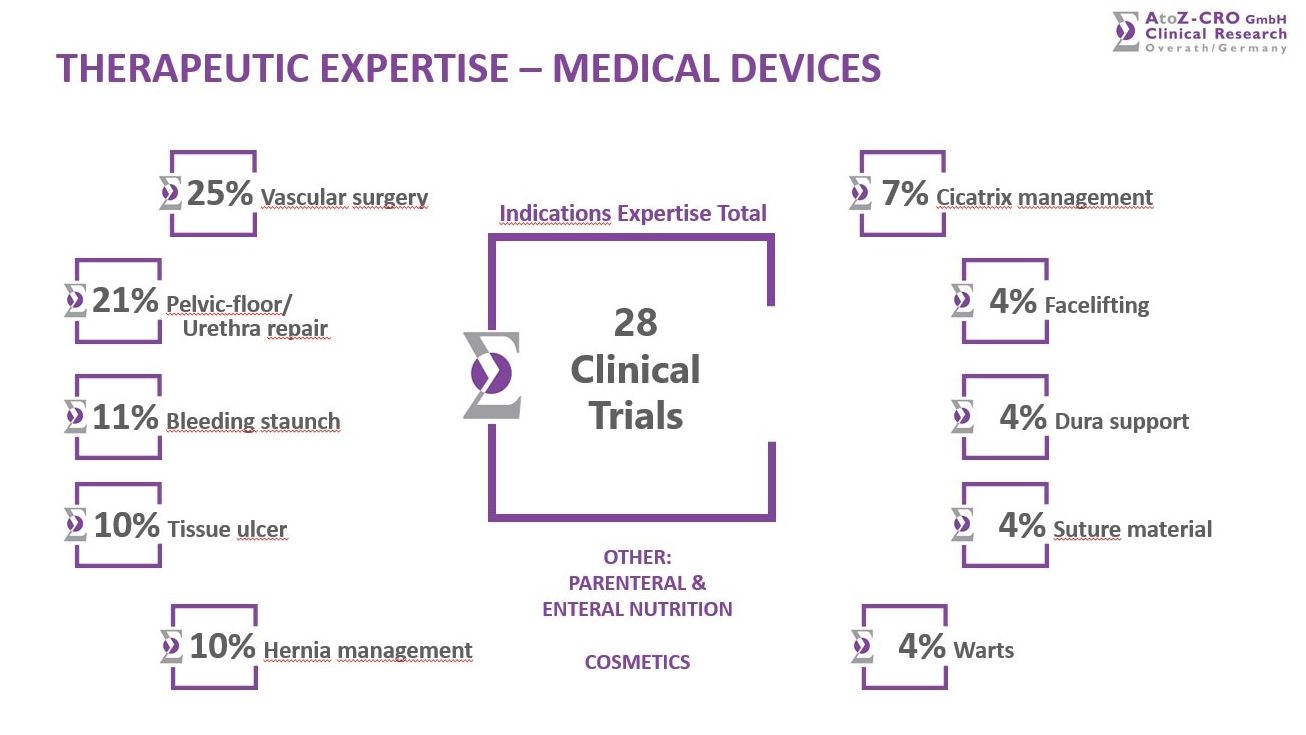
Amongst the 300+ performed studies we offer a very broad Therapeutic Expertise in the following areas:
Cardiovascular Infectiology
Hematology/Immunology Gastroenterology/Hepatology
Neurology/Psychiatry Endocrinology
Oncology Orthopedic Disorders
Respiratory Dermatology
Urology Ophthalmology
Analgesia/Anesthesia Other