China accepts CANbridge’s NDA for CAN108 to treat cholestatic pruritus in PFIC
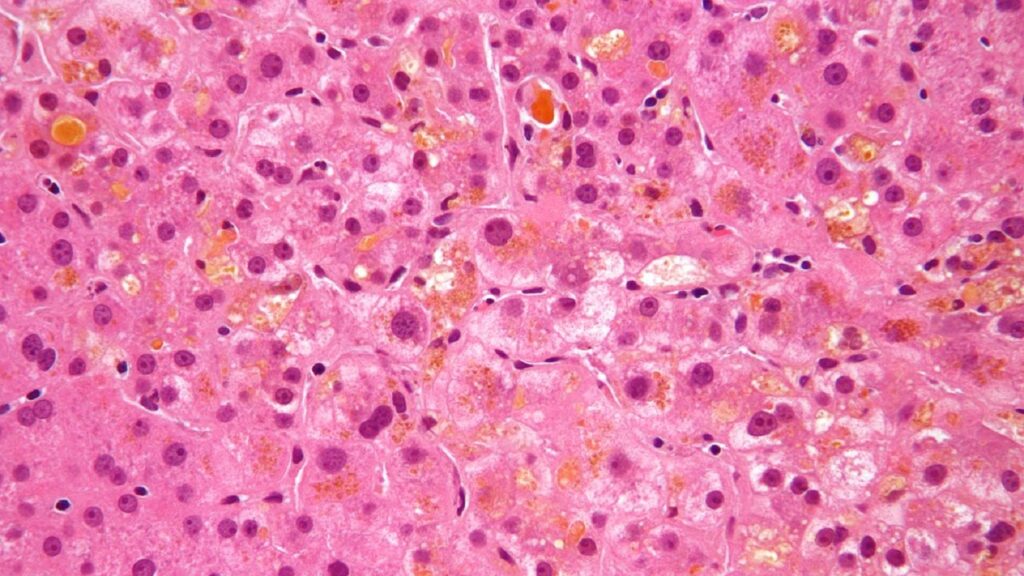
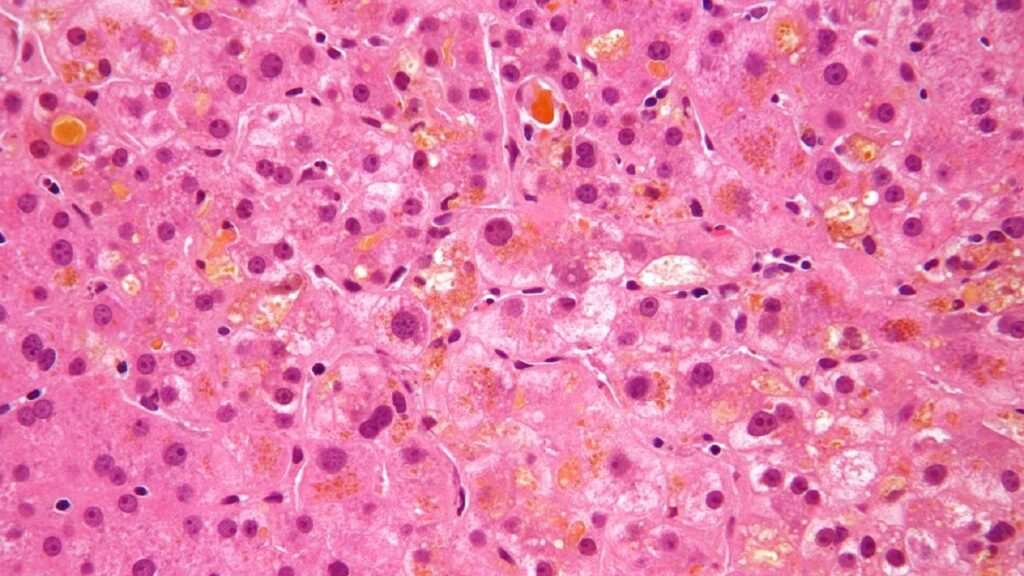
The National Medical Products Administration (NMPA) of China has accepted the new drug application (NDA) submitted by CANbridge Pharmaceuticals for CAN108 (Maralixibat Chloride Oral Solution/ LIVMARLI) for the treatment of cholestatic pruritus in patients aged two months and above with progressive familial intrahepatic cholestasis (PFIC).
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.