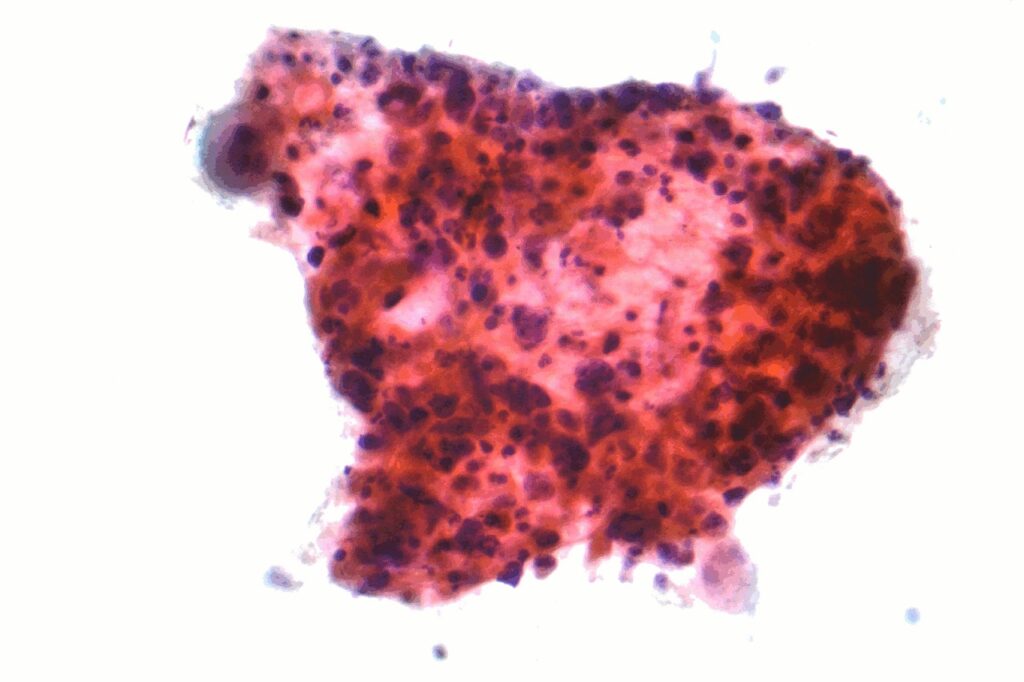
Glenmark gets DCGI nod to carry out Hodgkin’s lymphoma treatment trial
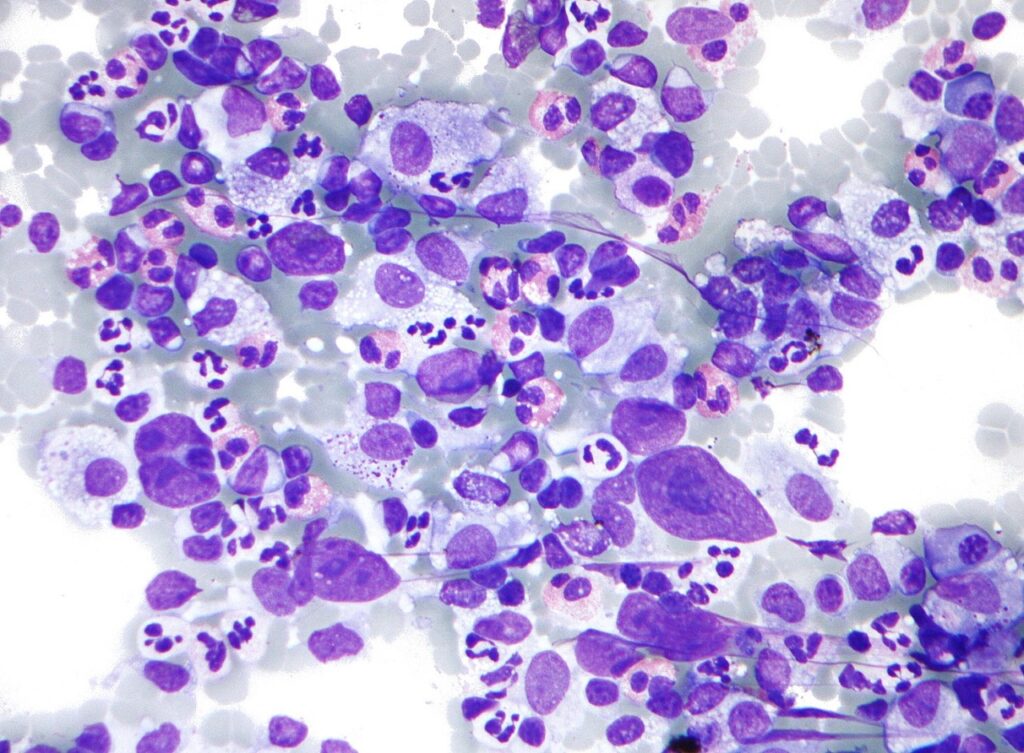
Glenmark Pharmaceuticals’ subsidiary Glenmark Specialty has received the Drugs Controller General of India (DCGI) approval to initiate the Phase I clinical trial of its new small-molecule, GRC 54276, in advanced solid tumours and Hodgkin's lymphoma patients.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.