Amicus Therapeutics has received positive opinion from the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) for Opfolda (miglustat) to treat late-onset Pompe disease (LOPD).
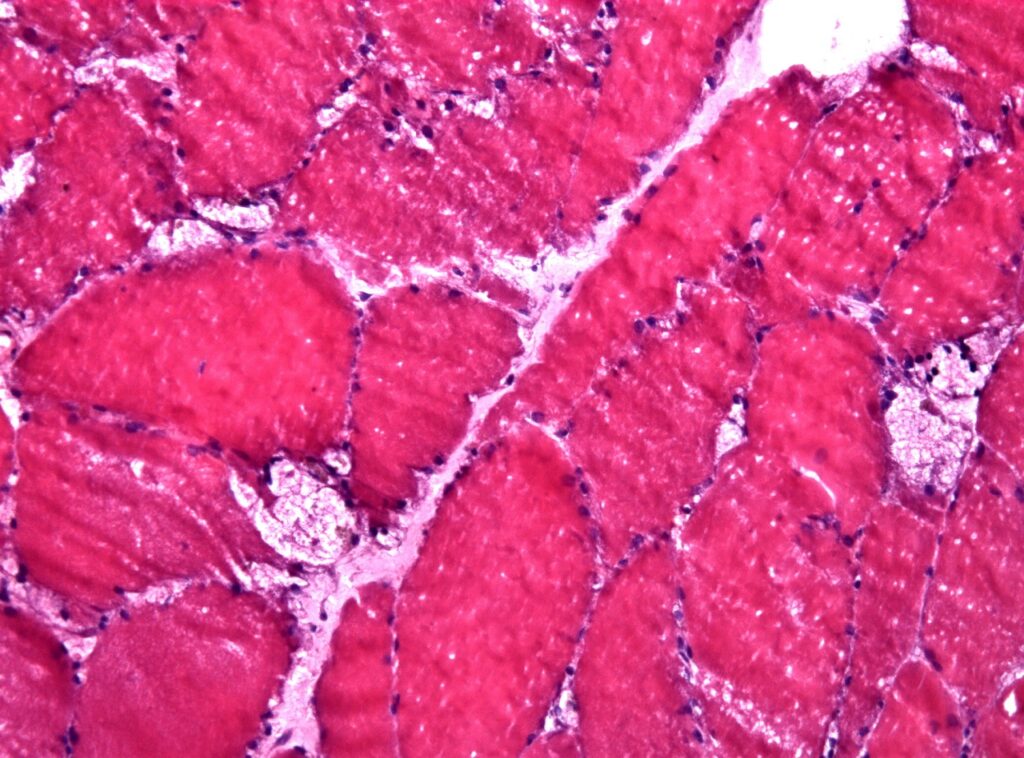
Muscle biopsy showing large vacuoles in a case of pompes disease. Credit: Jensflorian/ commons.wikimedia.org.
Subscribe to our email newsletter
Opfolda will be used as an enzyme stabiliser of cipaglucosidase alfa long-term enzyme replacement therapy in LOPD (acid α glucosidase [GAA] deficiency) adult patients.
The European Commission (EC) is expected to take decision on miglustat’s authorisation in the third quarter of this year.
Pombiliti (cipaglucosidase alfa), the biologic component of the two-component therapy, received EC approval last month.
Upon approval of Opfolda, Pombiliti + Opfolda, also called as AT-GAA, will become the first two-component therapy in the European Union (EU) to treat LOPD in adult patients.
Amicus Therapeutics executive chairman John Crowley said: “With today’s positive CHMP opinion of Opfolda, we are now one step away from bringing this much-needed new treatment to adults living in Europe with late-onset Pompe disease.”
The CHMP’s positive opinion was based on the clinical data obtained from the Phase III PROPEL pivotal study.
The randomised, controlled trial was conducted in LOPD patients who are ERT-naive and ERT-experienced.
Pombiliti + Opfolda comprises cipaglucosidase alfa, a bis-M6P-enriched rhGAA and the oral enzyme stabiliser, miglustat, which minimises the enzyme activity loss in blood.
Cipaglucosidase alfa has been designed to facilitate high-affinity uptake through the M6P receptor and retains its capacity to process into the most active form of the enzyme.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.