The China National Medical Products Administration (NMPA) has accepted Everest Medicines’ investigational new drug (IND) application for its selective immunoproteasome inhibitor, zetomipzomib.
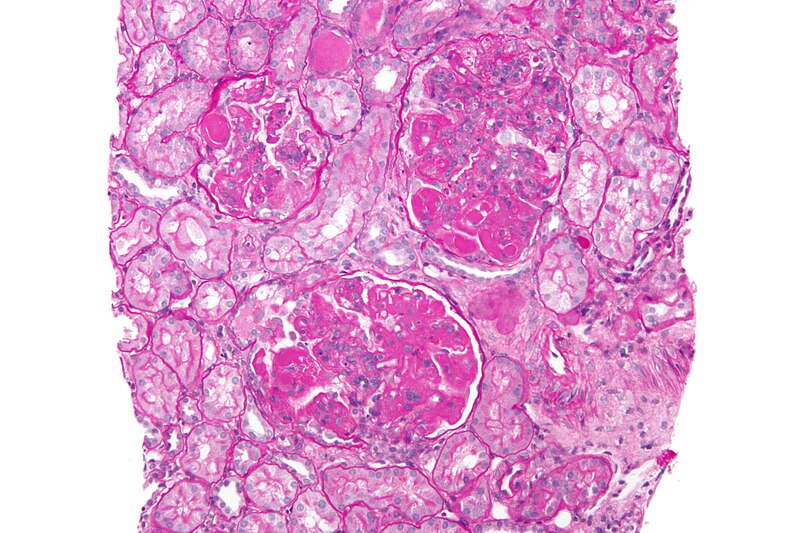
Lupus nephritis is the most common secondary immune-mediated glomerular disease. Credit: Nephron/commons.wikimedia.org.
Subscribe to our email newsletter
The new, first-in-class inhibitor is being assessed to treat a range of immune-mediated disorders, including lupus nephritis (LN), the most common secondary immune-mediated glomerular disease.
Following the acceptance, the company plans to join its partner, Kezar Life Sciences, for the international, placebo-controlled, randomised, double-blind Phase IIb PALIZADE study of zetomipzomib.
The trial expects to enrol 279 subjects, randomly assigned to receive 30mg, 60mg of zetomipzomib or placebo subcutaneously once-a-week for 52 weeks, besides standard background therapy.
It will assess the efficacy and safety of the two dose-levels of the therapy in active LN patients.
Everest Medicines CEO Rogers Yongqing Luo said: “The IND acceptance of zetomipzomib marks an important step towards entering clinical studies in China.
“We look forward to participating in the PALIZADE trial to enrol LN patients in the nation which has a high prevalence of the disease.
“Renal and autoimmune diseases are key therapeutic areas for Everest, and zetomipzomib represents another mid-to-late-stage pipeline asset that will solidify our leading position in these therapeutic areas in Asia.”
In an earlier Phase II study, the therapy demonstrated positive data, with clinically meaningful overall renal response rate of 64.7% at week 25 (end of treatment) and 88.2% at week 37.
At week 25, complete renal response rate was 35.3% and at week 37 it was 41.2%.
Zetomipzomib also demonstrated a favourable tolerability and safety profile during the trial and no new safety signals were reported during the follow-up period.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.