Eligo Bioscience has received Orphan Drug Designation (ODD) and Rare Pediatric Disease (RPD) designation from the US Food and Drug Administration (FDA) for its microbiome gene therapy, EB003.
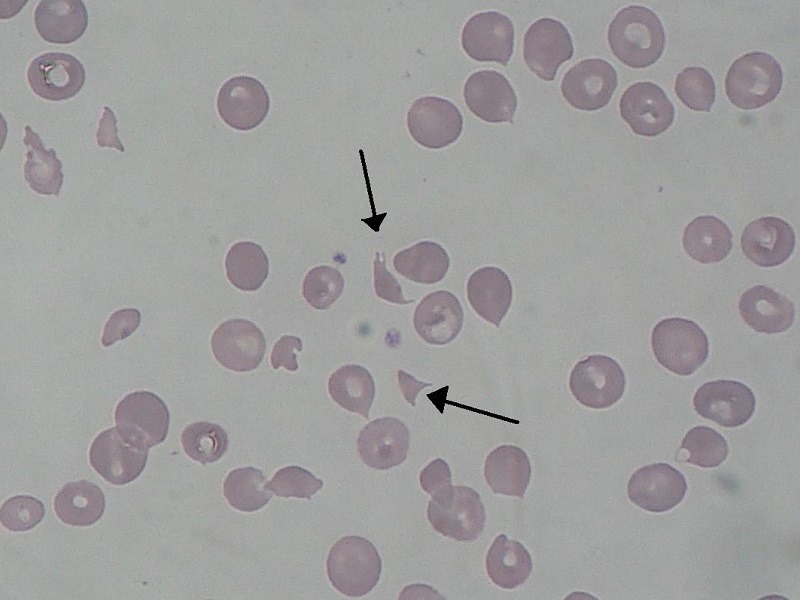
Schizocytes (hemolytic-uremic syndrome). Credit: Paulo Henrique Orlandi Mourao / commons.wikimedia.org.
Subscribe to our email newsletter
The CRISPR-based gene therapy has been indicated to treat Shiga-toxin producing bacterial infection to prevent hemolytic uremic syndrome (HUS).
It has been designed to target Shiga toxins from E. coli (STEC) bacteria in the gut of infected patients.
The company stated that the preclinical data supports the capacity of EB003 in efficiently eliminating Shiga-toxin genes from the gut of infected patients.
This will lead to quick decrease in toxin levels and the associated symptoms, as well as in preventing the evolution to HUS.
Eligo Bioscience CEO Xavier Duportet said: “Granting of Orphan Drug Designation and Rare Pediatric Disease designation for EB003 highlights the FDA’s recognition of the potential of how our unique CRISPR-based modalities can be used to address devastating diseases driven by the expression of bacterial genes, such as hemolytic uremic syndrome.
“We are grateful that the FDA is providing additional support for the development of therapies geared towards rare pediatric diseases and encourages us in our mission to propose highly innovative solutions to patients in need.”
According to the company, children aged less than five years are mainly sensitive to the expression of STEC bacteria after having contaminated foods.
The production of toxin in the person’s gut triggers bloody diarrhea, and its translocation and accumulation in the systemic compartment can lead to HUS, which affects kidneys and blood platelets.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.