Global health care company Fresenius Kabi has launched Stimufend (pegfilgrastim-fpgk), its pegfilgrastim biosimilar, for cancer patients in the US.
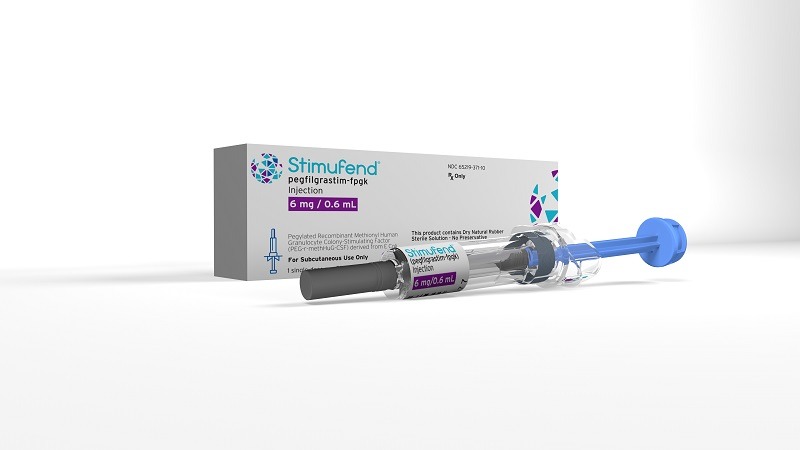
Stimufend is a leukocyte growth factor that is indicated for reducing the incidence of infection in patients with non-myeloid malignancies. Credit: Fresenius Kabi AG / Business Wire.
Subscribe to our email newsletter
In September last year, Stimufend received approval from the US Food and Drug Administration (FDA) to be used in patients with non-myeloid malignancies receiving myelosuppressive anti-cancer drugs that are associated with a clinically significant febrile neutropenia incidence.
Fresenius Kabi commercially launched Stimufend in a 6 mg/0.6 mL solution in a single-dose pre-filled syringe along with a passive needle guard.
The launch provides accessible and quality treatment option for cancer patients in the country who are undergoing chemotherapy.
Fresenius Kabi acting CEO Michael Sen said: “Fresenius Kabi’s first biosimilars launch in the US is a proud moment and a significant key milestone for us and our Vision 2026.
“We are expanding into a highly important and fast-growing market for biopharmaceuticals and Stimufend is the first of our biosimilars portfolio to support cancer patients in the US.
“Stimufend offers healthcare providers a high-quality treatment alternative while contributing to a sustainable healthcare system in the US.”
The company launched its pegfilgrastim biosimilar last October in a prefilled syringe in France after it secured marketing authorisation from the European Commission (EC) in March.
Stimufend is a leukocyte growth factor that is indicated for reducing the incidence of infection in patients with non-myeloid malignancies.
Fresenius Kabi chief operating officer Fresenius Kabi management board member Dr Michael Schönhofen said: “We are excited to provide this supportive care option to US patients and physicians via our dedicated US biosimilars organisation. This launch is a testimonial of our heritage in oncology and our commitment to oncology patients across the world.”
Stimufend is not indicated to mobilise peripheral blood progenitor cells for hematopoietic stem cell transplantation.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.