Mirati Therapeutics has received conditional marketing authorisation approval from the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA) for its advanced non-small cell lung cancer (NSCLC) therapy, Krazati (adagrasib).
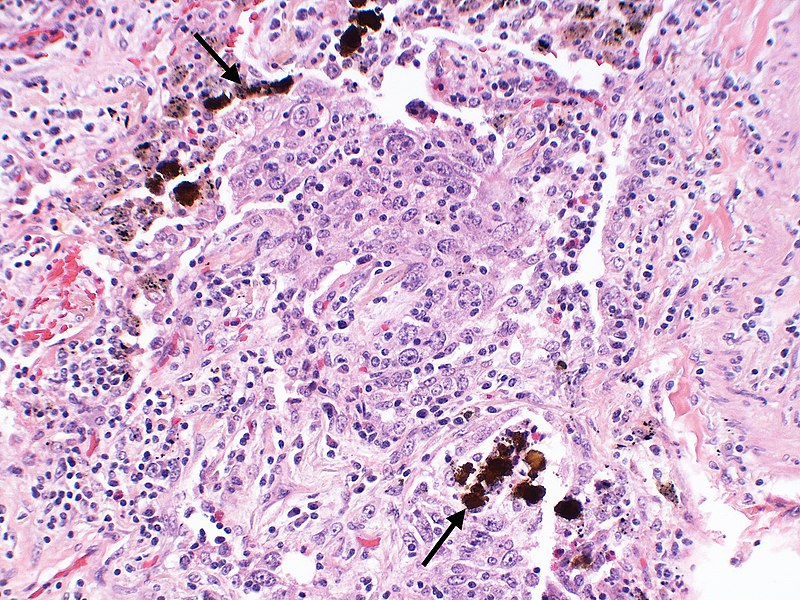
The black arrows point to ferrugionous bodies that are located at the periphery of a focus of non-small cell lung carcinoma, NOS. Credit: Yale Rosen from USA / commons.wikimedia.org.
Subscribe to our email newsletter
Krazati has been approved as a monotherapy to treat adult patients with advanced NSCLC with KRASG12C mutation and who have progressive disease after previous therapy with, or intolerance to, platinum-based chemotherapy and/or anti-PD-1/PD-L1 immunotherapy.
Adagrasib is designed to meet the challenge of KRASG12C by maintaining consistent target inhibition, which is vital for addressing KRASG12C-mutated cancers.
This is particularly important as the KRASG12C protein regenerates every 24-48 hours.
The therapy has demonstrated its ability to enter the central nervous system (CNS), a crucial factor considering the frequent occurrence of CNS metastases in NSCLC, which often leads to a poor prognosis.
The KRYSTAL-1 open-label Phase I/II multiple-expansion cohort study assessed adagrasib as a single agent and along with other anti-cancer therapies in patients with advanced solid tumours harbouring the KRASG12C mutation.
Mirati Therapeutics chief medical officer Alan Sandler said: “Krazati offers a compelling therapeutic option for patients with previously treated locally advanced NSCLC with a KRASG12C mutation.
“MHRA’s authorisation is a significant step towards improving the options available for patients and clinicians in Great Britain.”
Most lung cancers fall into two categories, including NSCLC, which constitutes around 85% of cases, and small cell lung cancer (SCLC), which makes up about 15% of cases.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.