BeiGene has received a positive recommendation from the UK’s National Institute for Health and Care Excellence (NICE) for Brukinsa (zanubrutinib) to treat adults with chronic lymphocytic leukaemia (CLL).
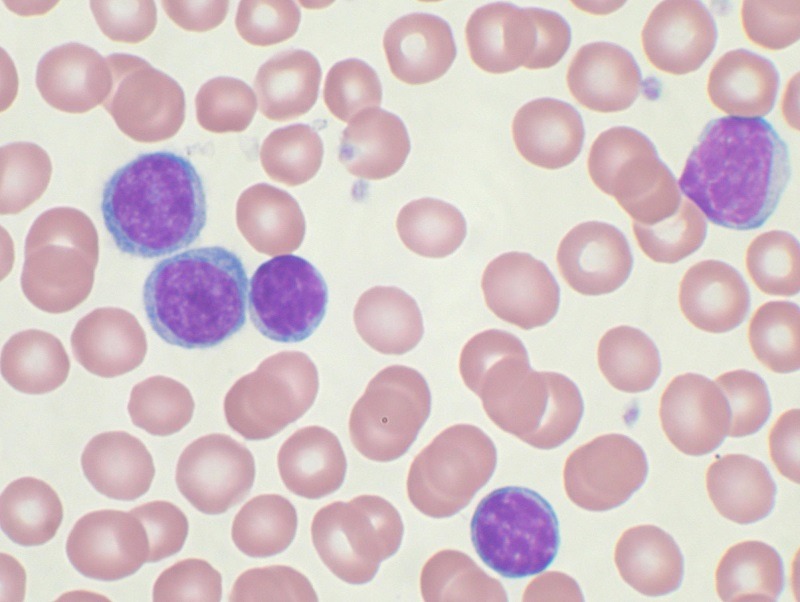
High-power magnification of a Wright's stained peripheral blood smear showing chronic lymphocytic leukaemia. Credit: Mary Ann Thompson / commons.wikimedia.org.
Subscribe to our email newsletter
NICE has released the final draft guidance (FDG) that suggests Brukinsa as a recommended treatment for eligible adults with untreated CLL if there is a 17p deletion or TP53 mutation.
It is also recommended for adults with untreated CLL without a 17p deletion or TP53 mutation, and fludarabine-cyclophosphamide-rituximab (FCR) or bendamustine plus rituximab (BR) is unsuitable, as well as relapsed or refractory CLL.
BeiGene UK and Ireland general manager Dr Robert Mulrooney said: “This follows the previous approval of Brukinsa by NICE in July 2022 as the only cost-effective treatment for patients with Waldenstrom’s macroglobulinemia.
“Although we are a relatively new player in the UK market, we are rapidly establishing ourselves as a company that can make innovative cancer medicines accessible and affordable for UK patients.”
Brukinsa, discovered by BeiGene scientists, is a small molecule inhibitor of Bruton’s tyrosine kinase (BTK).
It is currently undergoing worldwide clinical evaluation as a monotherapy and in combination with other therapies for a range of B-cell malignancies.
This therapy was designed to ensure continuous and effective inhibition of the BTK protein by optimising factors like bioavailability, half-life, and selectivity, as new BTK is consistently synthesized.
Brukinsa features differentiated pharmacokinetics compared to other approved BTK inhibitors.
The therapy has shown its effectiveness in inhibiting the growth of malignant B cells in various disease-relevant tissues.
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.