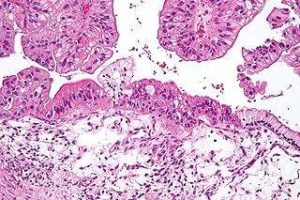
EMA accepts Clovis’ application for expanded indication of Rubraca to treat ovarian cancer
The regulator has accepted the application for a type II variation to the marketing authorization for Rubracato include maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.