Vertex Pharmaceuticals Incorporated (Nasdaq: VRTX) today announced that it has selected the triple combination of the next-generation corrector VX-445 (elexacaftor), tezacaftor and ivacaftor to submit for potential global regulatory approvals for people ages 12 and older with cystic fibrosis (CF).
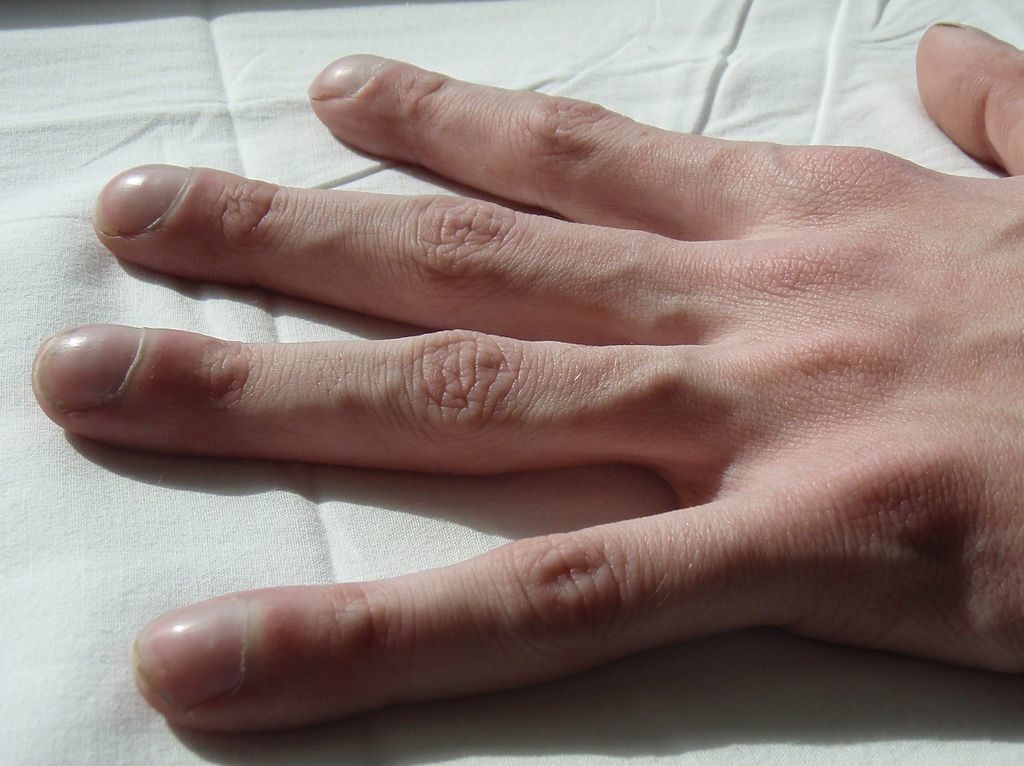
Image: Clubbing of the fingers is a classic feature of Cystic Fibrosis. Photo: courtesy of Jerry Nick, M.D./Wikipedia.org.
Subscribe to our email newsletter
Final data announced today from a 24-week Phase 3 study in people with one F508del mutation and one minimal function mutation and from a 4-week Phase 3 study in people with two F508del mutations will form the basis of these submissions.
Vertex previously announced that both of these Phase 3 studies met their primary endpoints, and the company today announced the final results of these studies, including results for key secondary endpoints and safety data.
In each study, treatment with the VX-445 triple combination regimen resulted in statistically significant improvements in all key secondary endpoints. Data from the 24-week study in people with one F508del mutation and one minimal function mutation showed a mean absolute improvement in percent predicted forced expiratory volume in one second (ppFEV1) of 14.3 percentage points from baseline (p<0.0001) and a 63% reduction in the annualized rate of pulmonary exacerbations (p<0.0001) through week 24 in patients who received the VX-445 triple combination regimen compared to those who received triple placebo. The VX-445 triple combination regimen was generally well tolerated across the two Phase 3 studies. In the study in people with one F508del mutation and one minimal function mutation, 2 and 0 patients, respectively, who received the VX-445 triple combination or triple placebo discontinued treatment due to adverse events. There were no discontinuations for adverse events in either arm of the study in people with two F508del mutations.
Vertex plans to submit a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) in the third quarter of 2019 and a Marketing Authorization Application (MAA) to the European Medicines Agency (EMA) in the fourth quarter of 2019 based on these data in people with CF ages 12 years and older who have one F508del mutation and one minimal function mutation and in people with two F508del mutations.
“The substantial improvements in lung function and other measures of CF seen in these Phase 3 studies are unprecedented and represent a defining moment in the journey to provide medicines that treat the underlying cause of CF to the vast majority of people with CF,” said Steven M. Rowe, M.D., M.S.P.H., Director of the Gregory Fleming James Cystic Fibrosis Research Center, University of Alabama at Birmingham and co-chair of Vertex’s Triple Combination Steering Committee.
“People with CF who have one F508del mutation and one minimal function mutation are the largest remaining group of CF patients without a treatment option for the underlying cause of their disease. The final Phase 3 data announced today represent a significant step toward bringing a disease modifying medicine to these patients, as well as toward providing significantly enhanced benefits to patients with two F508del mutations,” said Reshma Kewalramani, M.D., Executive Vice President, Global Medicines Development and Medical Affairs and Chief Medical Officer at Vertex. “We could not have achieved this important milestone without the support of the entire CF community, and we are particularly grateful to those who participated in the triple combination clinical trials. We now look forward to completing our regulatory submissions with the aim of bringing the VX-445 triple combination regimen to as many patients as possible.”
Vertex conducted two Phase 3 programs in parallel that evaluated two different triple combination regimens with the goal of selecting the best regimen to submit for global regulatory approvals. One program evaluated VX-659 in combination with tezacaftor and ivacaftor, and the other program evaluated VX-445 in combination with tezacaftor and ivacaftor. Each of the programs consisted of a Phase 3 study in people with one F508del mutation and one minimal function mutation and a Phase 3 study in people with two F508del mutations. The Phase 3 studies in both programs met their primary endpoints and showed statistically significant improvements across all key secondary endpoints. Additionally, each of the two triple combination regimens was generally well tolerated, and more than 98% of patients in each of the two programs completed the Phase 3 study treatment periods and elected to enroll in the open-label extension study for each regimen.
Both regimens showed highly similar and positive benefit-risk profiles. Vertex ultimately determined that the VX-445 triple combination regimen could benefit the greatest number of CF patients. This decision was based on a detailed assessment of multiple factors, including favorable profiles for safety, tolerability and drug-drug interactions, the ability for co-administration with hormonal contraceptives, and the lack of photosensitivity.
Source: Company Press Release
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.