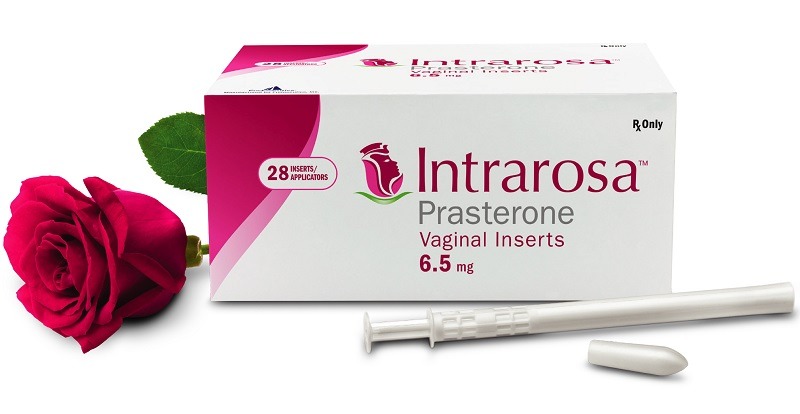
Cosette acquires worldwide rights to Endoceutics’ Intraros prescription drug
The transaction includes 108 issued and pending patents globally with the latest expiring this year including three Orange Book listed patents. The acquisition will expedite Cosette’s women’s health
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.