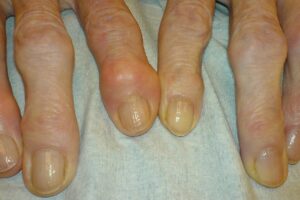
Levolta Pharmaceuticals has entered into an exclusive licensing and distribution agreement with Tabuk Pharmaceutical for commercialising VOLT01, an investigational osteoarthritis (OA) therapy in the Middle East and Africa (MEA) region.
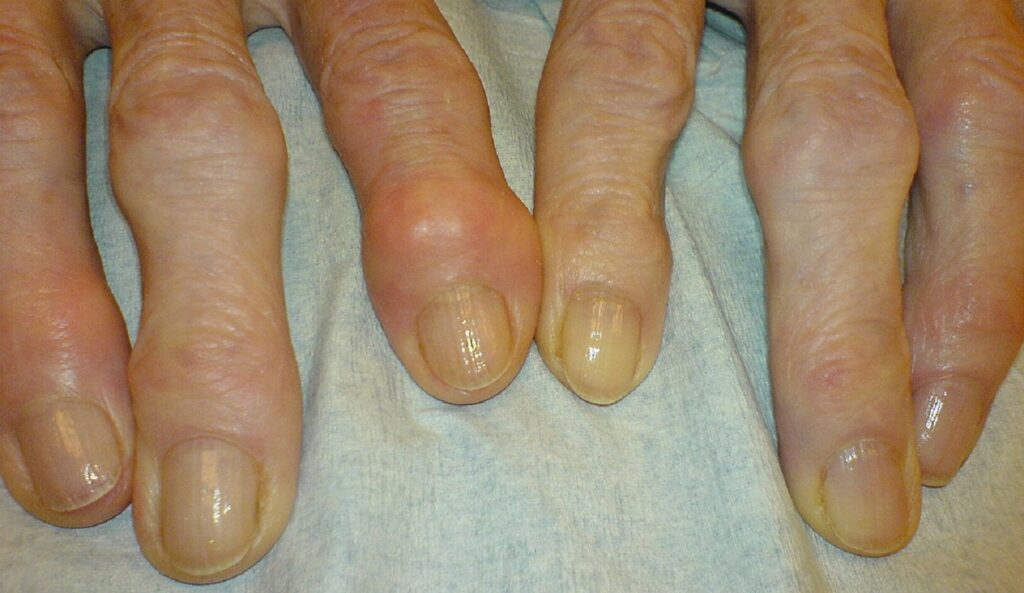
Tabuk will be responsible for registering, importing, and commercialising the OA therapy in the MEA region. Credit: Drahreg01 / commons.wikimedia.org.
Subscribe to our email newsletter
Tabuk is a fully owned subsidiary of Astra Industrial Group.
Under the agreement, Tabuk will hold the marketing authorisation and will be responsible for registering, importing, and commercialising the therapy in the MEA region, excluding South Africa.
Levolta will handle product manufacturing and quality assurance initiatives.
Levolta chief medical officer Ketan Desai said: “Our Phase II studies demonstrate long lasting pain relief, up to nine months, in patients with osteoarthritis after a single 30-minute intravenous infusion.
“More importantly, VOLT01 showed disease modifying activity as measured by MRI with pain relief correlating with disease modification.
“We look forward to pivotal studies that will enable VOLT01 to have a label for pain relief of osteoarthritis in all joints along with disease modification after a single yearly infusion.”
Levolta intends to initiate Phase III clinical trials for VOLT01 late this year or early next year.
Tabuk Pharmaceuticals CEO and Astra Industrial Group president Mohammed Al Hagbani said: “We are confident that VOLT01 will play a major role in improving the health condition of more than 5 million osteoarthritis patients in Saudi Arabia as well as many other patients across the region suffering from this disease.
“Our partnership with Levolta will strengthen our commitment to further support the Kingdom with the 2030 vision of providing unique medications addressing different therapy areas.”
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.