USFDA grants fast track designation for Kintara’s breast cancer therapy
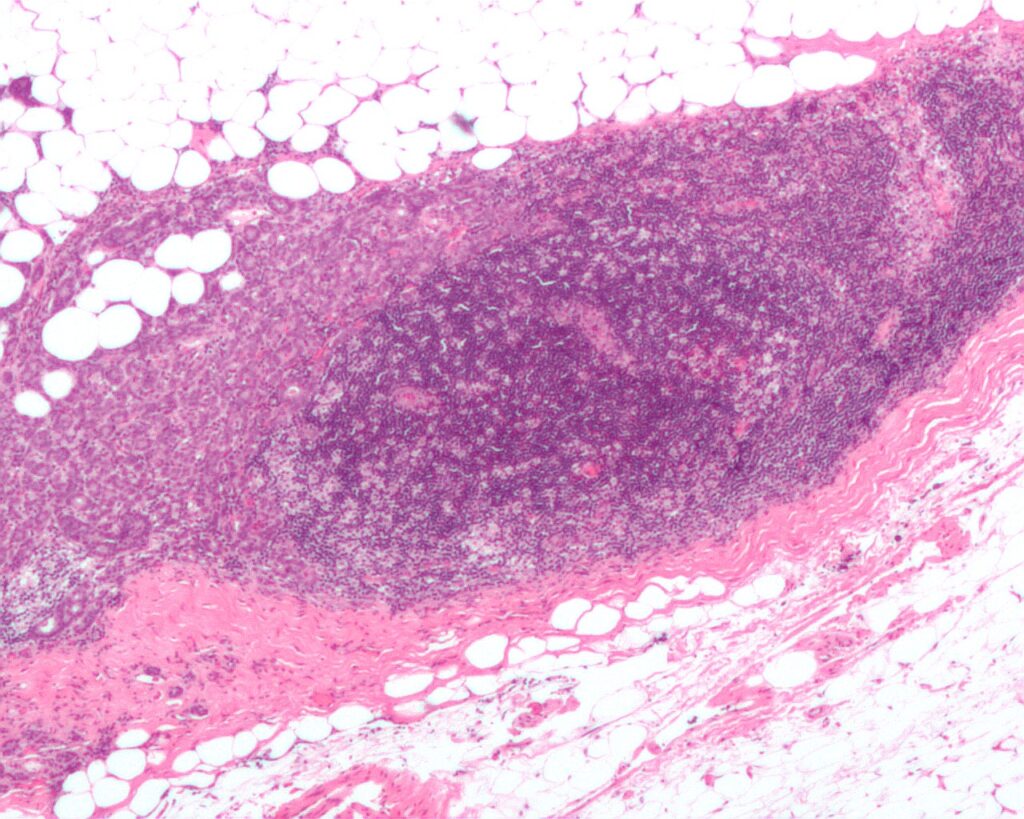
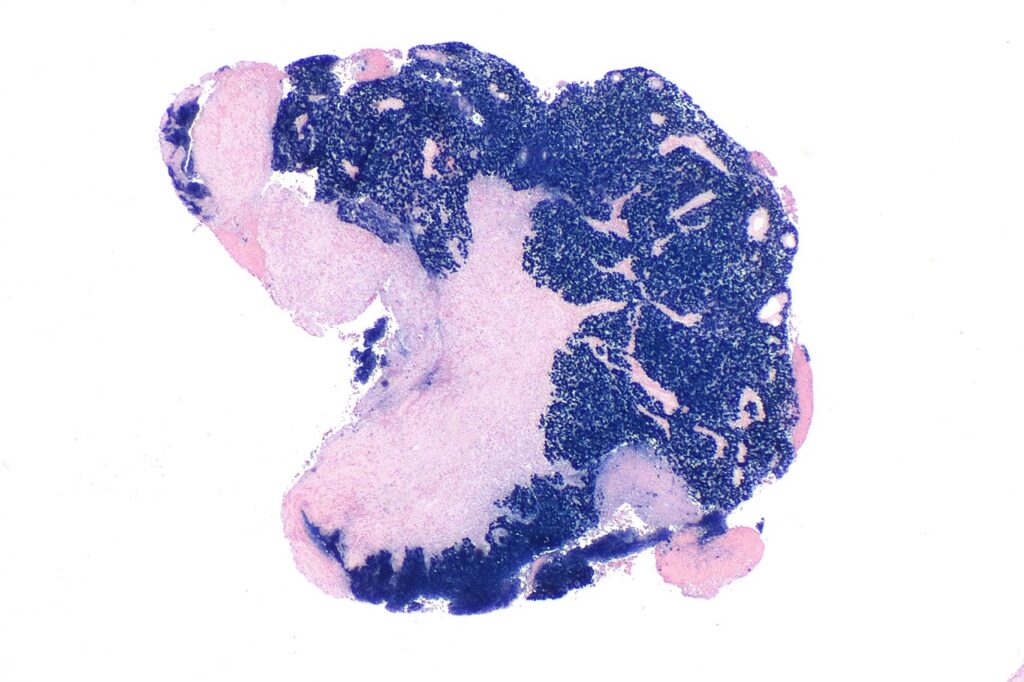
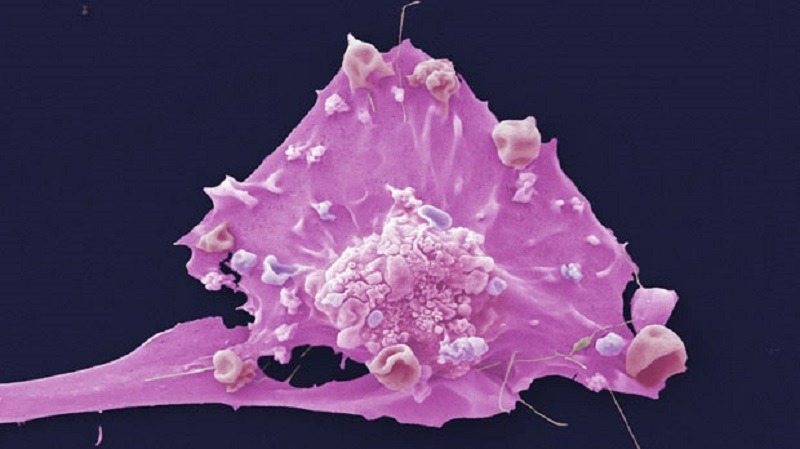
The US Food and Drug Administration (FDA) has granted fast track designation (FTD) for Kintara Therapeutics’ late-stage photodynamic therapy (PDT), REM-001, to treat cutaneous metastatic breast cancer (CMBC).
 Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
Advertise With UsAdvertise on our extensive network of industry websites and newsletters.
 Get the PBR newsletterSign up to our free email to get all the latest PBR
news.
Get the PBR newsletterSign up to our free email to get all the latest PBR
news.